ENERGY CHANGES IN CHEMICAL AND NUCLEAR REACTIONS
What are Energy Changes?
Energy changes are the transformations of energy from one form to another. The Law of Conservation of Energy states that energy cannot be created or destroyed; it is simply conserved from one form to another. Thus, we cannot say that energy is used up (ie. car uses up gas) but rather that the energy has changed (ie. chemical energy in gas has been change to thermal and kinetic energy in the car’s engine).
Energy changes are the transformations of energy from one form to another. The Law of Conservation of Energy states that energy cannot be created or destroyed; it is simply conserved from one form to another. Thus, we cannot say that energy is used up (ie. car uses up gas) but rather that the energy has changed (ie. chemical energy in gas has been change to thermal and kinetic energy in the car’s engine).
LESSON OVERVIEW (SPECIFIC EXPECTATIONS: D1.1, D2.1, D3.1, D3.2, D3.6)
|
Lesson Hook/Minds On:
Show students YouTube clip about the Fukushima Nuclear Reactor meltdown. This section compares chemical and nuclear reactions while also diving into very specific thermochemical concepts and ideas. There is often a lot of controversy and mystery surrounding nuclear reactions and power plants. Many people are unaware of what really goes in such a reaction and as such this video clip may be used to spark curiosity. To debrief the video, initiate a brief classroom discussion by asking students: Do you think we should or should NOT use nuclear energy? |
|
Diagnostic Activity:
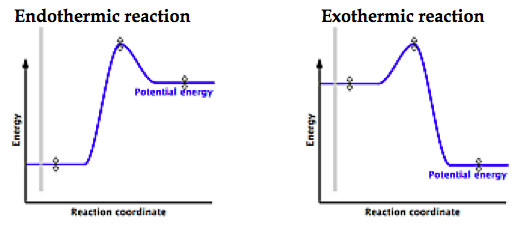
Gauge students’ incoming knowledge by playing a quick game of ‘exo vs endo’. Show students various pictures of reactions/heat transfer situations and ask them to decide whether they represent an endothermic or exothermic reaction. You could simply ask students to raise their hands for which option they think is correct. Another alternative for kinesthetic learners is to have a line down the middle of the class and designate one side as ‘exo’ and the other as ‘endo’. Students can then go to the side of the class they feel is correct. Finally, students can be asked to write in big letters on a piece of paper ‘endo’ and ‘exo’ and then raise the piece of paper to their foreheads so that only the teacher really sees the answers. To help ELL students, ‘exo’ can be colour coded as red and ‘endo’ as blue for example.
Gauge students’ incoming knowledge by playing a quick game of ‘exo vs endo’. Show students various pictures of reactions/heat transfer situations and ask them to decide whether they represent an endothermic or exothermic reaction. You could simply ask students to raise their hands for which option they think is correct. Another alternative for kinesthetic learners is to have a line down the middle of the class and designate one side as ‘exo’ and the other as ‘endo’. Students can then go to the side of the class they feel is correct. Finally, students can be asked to write in big letters on a piece of paper ‘endo’ and ‘exo’ and then raise the piece of paper to their foreheads so that only the teacher really sees the answers. To help ELL students, ‘exo’ can be colour coded as red and ‘endo’ as blue for example.
Teaching Strategy:
This section has very many terms and definitions that students will need to understand to grasp the material being presented. Students can be asked to make flash cards for all the key terms (as found in the ‘Key Terms and Concepts’ sub tab). The flash cards will be required to include a title, definition, example, and picture. The picture can be of a diagram where appropriate or simply tied to the example or definition. This activity will require students to engage with the textbook material or online resources to attain an in-depth comprehension of the key terms. The picture aspect can help visual learners, as well as give all students a better idea of energy change diagrams. This will be a great benefit for ELL students who can return to these flash cards throughout the unit and course to help solidify their understanding. Students could be motivated to do a good job by offering a quiz where students are allowed to bring their flash cards as an assist. Online flash card makers can speed up the process where computer, internet, and printing is available.
This section has very many terms and definitions that students will need to understand to grasp the material being presented. Students can be asked to make flash cards for all the key terms (as found in the ‘Key Terms and Concepts’ sub tab). The flash cards will be required to include a title, definition, example, and picture. The picture can be of a diagram where appropriate or simply tied to the example or definition. This activity will require students to engage with the textbook material or online resources to attain an in-depth comprehension of the key terms. The picture aspect can help visual learners, as well as give all students a better idea of energy change diagrams. This will be a great benefit for ELL students who can return to these flash cards throughout the unit and course to help solidify their understanding. Students could be motivated to do a good job by offering a quiz where students are allowed to bring their flash cards as an assist. Online flash card makers can speed up the process where computer, internet, and printing is available.
Consolidation:
After the activity, give students an exit card to be collected with the following question:
After gaining more information about nuclear reactions, do you think that we should or should NOT use nuclear energy? Give a reason as to why or why NOT from what you have learned today?
Students should still find the topic interesting albeit having been slightly more educated on it. By asking students to give a reason for their answer, they are connecting their thoughts and ideas to some of the concepts they have picked up in the lesson.
After the activity, give students an exit card to be collected with the following question:
After gaining more information about nuclear reactions, do you think that we should or should NOT use nuclear energy? Give a reason as to why or why NOT from what you have learned today?
Students should still find the topic interesting albeit having been slightly more educated on it. By asking students to give a reason for their answer, they are connecting their thoughts and ideas to some of the concepts they have picked up in the lesson.
FOPP: Commitment to students and student learning
Click below for a few resources that can be used in conjunction with a lesson to further demonstrate the concept of energy changes in chemical and nuclear reactions.