Quantum Numbers
4C
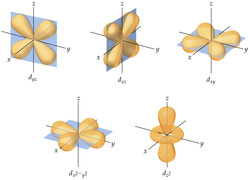
Scientists use mathematical equations to describe the energies of the electron in an atom, and the probability of finding the electron in a specific region. There are four main quantum numbers that describe the various types of orbitals. The principal quantum number (n) describes the size and energy of an atomic orbital. The secondary quantum number (l) describes the shape and energy of an atomic orbital. The magnetic quantum number (ml) describes the orientation of an atomic orbital in space. Lastly, the spin quantum number (ms) relates to the spin of the electron. Because students will be overwhelmed if presented all this information in one sitting, students will be using a jigsaw to learn the different numbers.
Hook:
Show a YouTube video about how MRI machines work. This will introduce them to the idea that electrons are constantly spinning in different directions.
Action:
The class is split up into groups of four, with a special attention to making sure that all groups work well together. Each person from the group joins a different master group in which they learn about one of the four quantum numbers. There will be iPads at each master group as they learn about the quantum number.
After, they will return to their home group and share what they learn with other group members.
After, they will return to their home group and share what they learn with other group members.
Consolidation:
The teacher uses questions to test the learning of the students, and collects the answers as an exit slip to ensure that students have the right information.
