Reaction Rates
What are Reaction Rates?
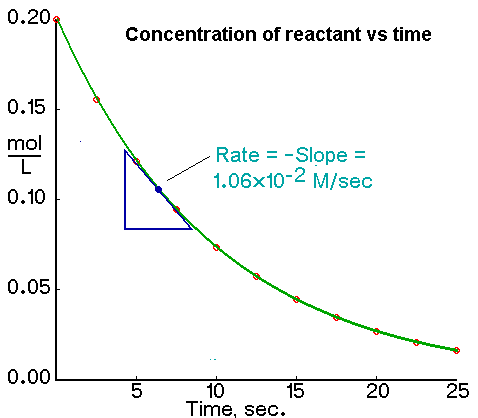
As a reaction progresses, the change over time in the concentration of a reactant or product is measured in what we call a 'reaction rate'. It can be measured both through calculations as well as graphically. This falls under the study of chemistry known as chemical kinetics.
adapted from:
DiGiuseppe, M., Haberer, S., Salciccioli, K., Sanader, M., & Vavitsas, A. (2012). Nelson Chemistry 12 University preparation. Toronto, ON: Nelson Education Ltd.
As a reaction progresses, the change over time in the concentration of a reactant or product is measured in what we call a 'reaction rate'. It can be measured both through calculations as well as graphically. This falls under the study of chemistry known as chemical kinetics.
adapted from:
DiGiuseppe, M., Haberer, S., Salciccioli, K., Sanader, M., & Vavitsas, A. (2012). Nelson Chemistry 12 University preparation. Toronto, ON: Nelson Education Ltd.
LESSON OVERVIEW (SPECIFIC EXPECTATIONS: D2.7, D3.4)
Lesson Hook/Minds On:
Perform mini lab demo/investigation using glow sticks. One set of glow sticks will be placed in hot water and one will be placed on ice. Next ask the students : "How does a glow stick turn on?"
Now ask students turn on a hot glow stick and a cold one and compare the differences. Finally ask students:
"Why do you think there is a difference in the brightness of the glow sticks?"
Perform mini lab demo/investigation using glow sticks. One set of glow sticks will be placed in hot water and one will be placed on ice. Next ask the students : "How does a glow stick turn on?"
Now ask students turn on a hot glow stick and a cold one and compare the differences. Finally ask students:
"Why do you think there is a difference in the brightness of the glow sticks?"
|
Teaching Strategy:
Students will conduct a lab activity to measure reaction rates. Students will carry out a reaction and track the changes of products versus reactants throughout. There are several follow up questions included to help guide student learning. Students can be placed in groups to facilitate each others learning, especially for interpersonal learners. The follow up questions can be completed for homework if there is not enough time in class. |
|
Consolidation:
As a consolidation, post on the board for students the following three topics: average reaction rates, instantaneous reaction rates, and stoichiometric rate relationships. Have students explore these topics and write brief definitions with examples using their textbooks or the internet. To be taken up next class. The link to the right is a webpage with thorough explanation with videos of the topics students are being asked to looked into. Students can be directed here or instructors can use this as their own resource.
As a consolidation, post on the board for students the following three topics: average reaction rates, instantaneous reaction rates, and stoichiometric rate relationships. Have students explore these topics and write brief definitions with examples using their textbooks or the internet. To be taken up next class. The link to the right is a webpage with thorough explanation with videos of the topics students are being asked to looked into. Students can be directed here or instructors can use this as their own resource.
FOPP: Professional practice
Click below for a few resources that can be used in conjunction with a lesson to further demonstrate the concept of reaction rates.