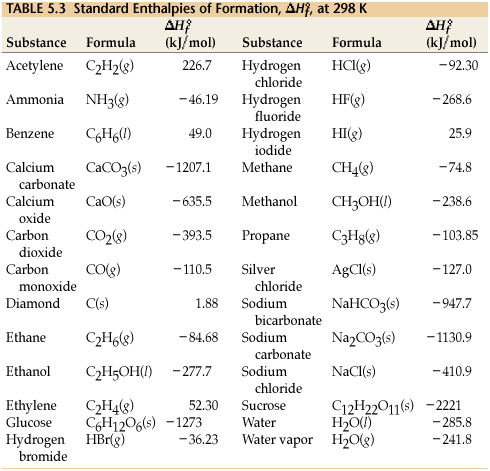
Standard Enthalpies of Formation
What are Standard Enthalpies of Formation?
When making a compound from its elements at standard state, there is an accompanying energy change. The enthalpy change needed to make 1 mole of our desired product is known as the standard enthalpy of formation.
adapted from:
DiGiuseppe, M., Haberer, S., Salciccioli, K., Sanader, M., & Vavitsas, A. (2012). Nelson Chemistry 12 University preparation. Toronto, ON: Nelson Education Ltd.
When making a compound from its elements at standard state, there is an accompanying energy change. The enthalpy change needed to make 1 mole of our desired product is known as the standard enthalpy of formation.
adapted from:
DiGiuseppe, M., Haberer, S., Salciccioli, K., Sanader, M., & Vavitsas, A. (2012). Nelson Chemistry 12 University preparation. Toronto, ON: Nelson Education Ltd.
LESSON OVERVIEW (SPECIFIC EXPECTATIONS: D2.7, D3.4)
Lesson Hook/Minds On:
As students enter class, have them write the number of a question from the previous day’s homework on Hess’ Law calculations that they found difficult. Tally up which question most students found challenging and have students come up and attempt to solve it for everyone with guiding tips from peers as well as the teacher.
As students enter class, have them write the number of a question from the previous day’s homework on Hess’ Law calculations that they found difficult. Tally up which question most students found challenging and have students come up and attempt to solve it for everyone with guiding tips from peers as well as the teacher.
Teaching Strategy:
To help teach the concept of standard enthalpies of formation, you most first model for students how to solve a few sample problems. Next pair off students and conduct a Practice Problem ‘Space Race’ via Socrative. Using any internet enabled device; students will view the problem, have to solve it, and submit the answer online. There more correct answer they have, the further their teams space ship flies. First team to the finish wins (a prize maybe for added incentive?).
The link below provides a website with several sample problems and solutions.
To help teach the concept of standard enthalpies of formation, you most first model for students how to solve a few sample problems. Next pair off students and conduct a Practice Problem ‘Space Race’ via Socrative. Using any internet enabled device; students will view the problem, have to solve it, and submit the answer online. There more correct answer they have, the further their teams space ship flies. First team to the finish wins (a prize maybe for added incentive?).
The link below provides a website with several sample problems and solutions.
Consolidation:
Using the real time results of the space race, see which questions students had struggled most with and go over them step by step to clarify.
Using the real time results of the space race, see which questions students had struggled most with and go over them step by step to clarify.
|
The button below links to Socrative, the online questioning and response system.
To the right is a table of Standard Enthalpies of Formation that students will need to solve the practice problems. |
FOPP: Leadership in learning communities
Click below for a few resources that can be used in conjunction with a lesson to further demonstrate the concept of standard enthalpies of formation.