Structure and Properties of Solids
13C
We know that certain solids are much harder than other solids. This topic seeks to explain why we observe these differences. There are four types of solids: ionic crystals, metallic crystals, molecular crystals, and covalent network crystals.
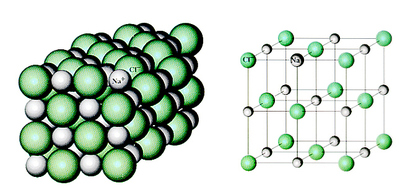
Ionic crystals form when a metal reacts with a non-metal to produce oppositely charged ions that are attracted to each other.
Metallic crystals are bound by a sea of electrons.
Molecular crystals are in part determined from the intermolecular dipole-dipole and hydrogen bonds.
Ionic crystals form when a metal reacts with a non-metal to produce oppositely charged ions that are attracted to each other.
Metallic crystals are bound by a sea of electrons.
Molecular crystals are in part determined from the intermolecular dipole-dipole and hydrogen bonds.
Hook:
Rockwell scale shows the hardness of different substances.
Action:
Students complete will complete a Venn diagram comparing the various properties of different kinds of solids.
Consolidate:
Students will complete post the Venn diagrams and complete a gallery walk. For additional practice, questions #1-6, in Nelson Chemistry 12 on page 254.
