THE RELATIONSHIP BETWEEN CHEMICAL AND ELECTRICAL ENERGY
|
It has been known for a very long time, before the 1800s, that the friction of two objects with one another produced static electricity. However, scientists then could not take advantage of the electricity produced by friction, as it seemed that the sparks produced could not be put to use. Alessandro Volta, pictured below, invented the electric cell, and by doing so, paved the way for the development of many practical ways of utilizing electricity.
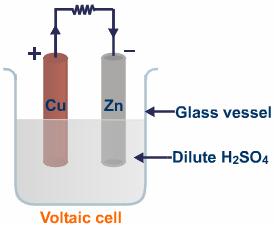
Although Volta is credited with inventing the first electric cell, it is a known fact that his inspiration was the work if an Italian biologist called Luigi Galvani (pictured on the left). Galvani studied the effects of electric current on the nerves, something that legend has it, he accidentally discovered. Over time, the principles behind Galvani’s observations have allowed scientists to develop what we know as “batteries.” As it is commonly known, batteries come in all sizes and shapes, and the function of many objects depends on the correct functioning of batteries. An example of this is a pacemaker. Needles to say, it is imperative that a pacemaker’s battery function properly. It is estimated that more than $50 billion a year is spent in the battery industry, either to replace them, or to insure that equipment which depends on the life of batteries is properly working. It is important, for our purposes, that we teach our students that regardless of the complexity of their design, all batteries have the same basic components, that being two different substances undergoing a redox reaction, and an electrolyte. These are all concepts that will be expanded throughout the following 5 lessons. |
References
- Image on top retrieved from: http://www.blueplanetgreenliving.com/wp-content/uploads/2008/11/duracell_575.jpg
- Di Guiseppe, M., Haberer, S., Salciccioli, K., Sanader, M, Vavitsas, A. (2012) Chemistry 12. Toronto: Nelson Education Ltd.
- van Kessel, H., Jenkins, F., Davies, L., Plumb, D., Di Guiseppe, M., Lantz, O., Tompkins, D. (2003). Chemistry 12. Toronto: Thomson Canada Limited