Common Misconceptions
Some of the topics covered in thermochemistry and chemical kinetics can be quite abstract for students in grade 12 university chemistry to understand. Below are some misconceptions students may struggle with along with clarifications and activity suggestions.
1. Students often think that energy is released when we break bonds.
Students may think that fuels store chemical energy and then release it in combustion. They might think that the fuel is being broken into smaller molecules in combustion, and that the breaking of the bonds holding the fuel molecules together is what releases the energy. To clarify: energy needs to be absorbed to break chemical bonds and it is released when new bonds form. So in combustion, the amount of energy released to form the bonds in the products is GREATER than the amount of energy needed to break the bonds in the reactants. Hence there is a net release of energy.
An activity to help students overcome this misconception involves rubber bands. Students can hold a rubber band in both hands. When they stretch their hands apart, they will see it is not easy and requires energy. When they bring their hands back together, the rubber band loosens and it is a lot easier. The same applies for molecules making and breaking bonds.
An activity to help students overcome this misconception involves rubber bands. Students can hold a rubber band in both hands. When they stretch their hands apart, they will see it is not easy and requires energy. When they bring their hands back together, the rubber band loosens and it is a lot easier. The same applies for molecules making and breaking bonds.
2. Students may think that energy is released when molecules no longer need to be held together.
Bond energies are characterized as quantities, which can further propagate the misconception that chemical energy is stored in bonds. Students may think that when molecules no longer need to be held together, that their stored energy is released. Again, energy is released when bonds form, and it is needed to break bonds. When bonds form it means the atoms are achieving a more stable configuration so they do not need energy to hold them together.
The rubber band activity from above is again applicable to help clarify this misconception for students. |
3. Students may think that alternative energy sources are ALWAYS the BEST.
Students may think that alternative energy sources are always better than traditional energy sources and should completely replace them right away. All energy sources have both costs and benefits. You must consider: how much money it costs, how it affects the environment, how reusable it is, how common and widespread it is, and how it can be used with today’s technology.
For an activity to help with this misconceptions, students can be divided into groups and each group will be assigned a different alternative / traditional source of energy that they will research. Students will construct a mind map of their topic in a communal class ‘Energy Sources’ mind map. This will all be done online through Prezi.
For an activity to help with this misconceptions, students can be divided into groups and each group will be assigned a different alternative / traditional source of energy that they will research. Students will construct a mind map of their topic in a communal class ‘Energy Sources’ mind map. This will all be done online through Prezi.
4. Students may think that wind energy is completely free.
Because we always have wind on Earth and it happens naturally, students may think that getting energy from wind is practically free. It can be explained to students that to get wind energy we must spend money to set up the windmills. These windmills can affect animals in the ecosystem, cause noise pollution, and might not be visually appealing (ie. would you want one in your backyard?).
Again, the alternative energy research activity can be used to clarify this misconception. |
5. Students may think that whether or not a reaction reaches completion is determined by the rate of the reaction.
Students may have trouble distinguishing that the rate of the reaction only indicates the speed with which it occurs and not how much reactant is used up in the process. To clarify, it can be explained that some very quick reactions appear to reach completion even after only a little portion of the reactants have been used. At the same time, very slow reactions might eventually turn all of the reactants into products.
A suggestion to help students clarify this misconception is to have them make a t-chart and list a few distinguishing points under each heading of: 'reaction rate' and 'reaction completion'. The link below takes you to a webpage with a large assortment of graphic organizers that can be printed for students.
A suggestion to help students clarify this misconception is to have them make a t-chart and list a few distinguishing points under each heading of: 'reaction rate' and 'reaction completion'. The link below takes you to a webpage with a large assortment of graphic organizers that can be printed for students.
6. Students may think that increasing the temperature lowers the activation energy.
Students may think that increasing the temperature speeds up the reaction by lowering the barrier of activation energy. Temperature like concentration, surface area, and the nature of reactants affect the rate of the reaction by changing the amount of collisions that have enough energy to overcome the activation energy barrier. Decreasing activation energy is done by catalysts that offer a lower energy pathway for the reaction to occur.
Showing students the 'Collision Theory Rap' is a fun way for students to grasp a better handle on the concept of collision theory. |
|
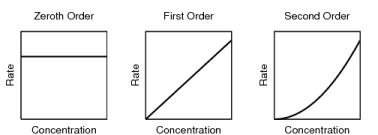
7. Students may think that the higher the rate law exponent, the higher the amount of reactant consumed.
|
Students may think that if more reactants are used up in the reaction, there will be a greater rate law exponent. In fact, the exponent is related to progression of the reaction through the types and number of collisions. In some reactions, the concentration of a reactant may have no effect on the rate of the reaction. These are called zero-order reactions. Measuring an individual reactant's effect of concentration on the reaction is the only way of obtaining the reaction order for said reactant.
Using the graphs can be a good way to help students connect the mathematical calculations to a visual representation of the processes taking place in chemical reactions. |
adapted from:
Nelson Chemistry 12 University Preparation
DiGiuseppe, M., Haberer, S., Salciccioli, K., Sanader, M., & Vavitsas, A. (2012). Nelson Chemistry 12 University preparation. Toronto, ON: Nelson Education Ltd. Nelson Chemistry 12 University Preparation Teacher's Resource
DiGiuseppe, M., & Sanader, M. (2012). Nelson Chemistry 12 University preparation Teacher's resource. Toronto, ON: Nelson Education Ltd.
Nelson Chemistry 12 University Preparation
DiGiuseppe, M., Haberer, S., Salciccioli, K., Sanader, M., & Vavitsas, A. (2012). Nelson Chemistry 12 University preparation. Toronto, ON: Nelson Education Ltd. Nelson Chemistry 12 University Preparation Teacher's Resource
DiGiuseppe, M., & Sanader, M. (2012). Nelson Chemistry 12 University preparation Teacher's resource. Toronto, ON: Nelson Education Ltd.