Intermolecular Forces
12C
Intermolecular forces refer to the chemical bond within a molecule. Van der Waals forces includes many types of intermolecular forces such as dipole-dipole forces, London dispersion forces, and hydrogen bonding. Using the knowledge of different types of forces, different properties can be predicted for compounds and elements.
Hook:
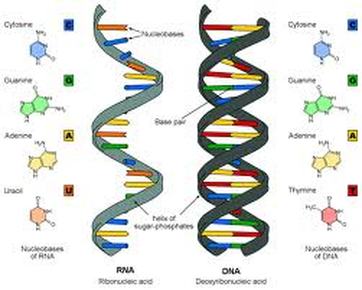
Discuss the double helix structure of DNA and how hydrogen bonding occurs between the rungs of the ladder.
Action

http://iosnova.com/ipad-mini-specs-and-information/
Teacher will use a jigsaw learning strategy and each group will learn about a different kind of intermolecular forces, and then return to their master group. Once again the different groups will have iPads or may use their PEDs to learn about one the aforementioned intermolecular forces. They will then return to their master group and share what they have learned.
Consolidation:
Students create a placemat of their findings. PED can be used to take a picture of placemat, and it can be uploaded to the class website.
