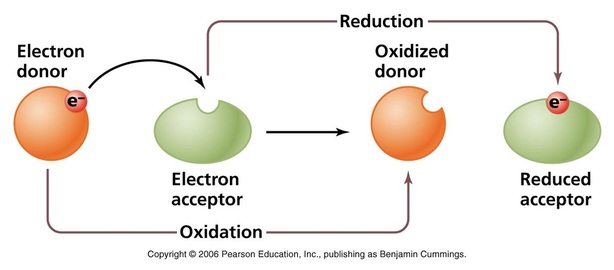
BALANCING REDOX REACTIONS
There are two basic methods for balancing redox equations. Oxidation numbers are a bookkeeping device that can be used to express oxidation states, to determine whether a reaction is redox or not, to determine which entity is oxidized and which is reduced, to determine which entities are the oxidizing and reducing agents, to determine how many electrons are exchanged, and to balance a redox equation. Although there are several oxidation-number methods for balancing redox equations, they all provide the same answer.
Half-reactions provide another way of keeping track of electron transfer in redox reactions. Both methods have one thing in common: that electrons gained and lost are the same, and that atoms are neither destroyed nor created.
Half-reactions provide another way of keeping track of electron transfer in redox reactions. Both methods have one thing in common: that electrons gained and lost are the same, and that atoms are neither destroyed nor created.
Learning Objectives
By the end of this lesson, I will:
· Be able to balance redox equations using the oxidation numbers method.
· Be able to balance redox equations using the half-reactions method.
· Be able to balance redox equations using the oxidation numbers method.
· Be able to balance redox equations using the half-reactions method.
Lesson Approaches and Overview (Specific Expectations: F2.1, F2.2, F2.3, F3.1)
· As a hook or introduction, the teacher will demonstrate the combustion of methane gas bubbles. The reference for this experiment is found here:
Please know that it is the teacher's responsibility to set and perform the lab, using the video as a reference.
· Referring back to this activity, introduce half reactions to introduce the balancing of redox equations.
· Referring back to this activity, introduce half reactions to introduce the balancing of redox equations.
· Use the following analogy to explain that all redox reactions are electron transfer reactions: “When you give a friend $5, you lose $5 and your friend gains $5, that is, $5 have been exchanged. Obviously, the money lost must equal the money gained. The same is true for electrons in a redox reaction.
· The following PowerPoint presentation can be used to start the explanation of redox equation balancing:
· The following PowerPoint presentation can be used to start the explanation of redox equation balancing:
· Explain that the definitions of oxidation and reduction in terms of change in oxidation number are consistent with the definitions in term of gain and loss of electrons. A reduction in oxidation number is reduction; an increase in oxidation number is oxidation.
· Require your students to draw an arc line to join the “atoms” of the same element with different oxidation states. Have students label the substances oxidized (LEO) and reduced (GER) and the substances that are the strongest oxidizing (SOA) and reducing agents (SRA).
· The following video can be used to explain the balancing of redox equations using the oxidation number method:
· Require your students to draw an arc line to join the “atoms” of the same element with different oxidation states. Have students label the substances oxidized (LEO) and reduced (GER) and the substances that are the strongest oxidizing (SOA) and reducing agents (SRA).
· The following video can be used to explain the balancing of redox equations using the oxidation number method:
Using the video as a reference, accompanied by a lecture that highlights the oxidation number method, ask students to create a flow chart to show the steps required to balance a redox equation using oxidation numbers.
· The following worksheet provides a clear and succinct summary of the half-reaction equation method:
· The following worksheet provides a clear and succinct summary of the half-reaction equation method:
· Use the following video can be used to introduced the balancing of redox equations using the half-reaction method in an acidic solution:
For the half-reaction equation method in a basic solution, use the following video:
· It is a good idea to go through the examples of writing complex half-equations in detail, modeling the steps required and showing the cancellation of the equal amount of identical entities. Visual learners will benefit by making and discussing a flow chart of the steps involved in writing complex half-reaction equations.
· It is important to highlight the similarities and differences between the acidic and basic half-reaction methods.
· When balancing half-reaction equations, students can use the memory aid “Major OH-“ to describe the order of steps to be used. “major OH-“ means balance major atoms first, then oxygen atoms, hydrogen atoms, and finally charge, using electrons.
· For the success of this lesson, it is important that students practice as much as they can. The teacher should provide students with worksheets to practice, as well as encourage students to do the problems on the textbook used.
· The following lab, a titration, is a good activity that incorporates lab procedures with the balancing of redox equations. Note that the teacher is responsible for supplying all materials needed, as well as supervising:
· It is important to highlight the similarities and differences between the acidic and basic half-reaction methods.
· When balancing half-reaction equations, students can use the memory aid “Major OH-“ to describe the order of steps to be used. “major OH-“ means balance major atoms first, then oxygen atoms, hydrogen atoms, and finally charge, using electrons.
· For the success of this lesson, it is important that students practice as much as they can. The teacher should provide students with worksheets to practice, as well as encourage students to do the problems on the textbook used.
· The following lab, a titration, is a good activity that incorporates lab procedures with the balancing of redox equations. Note that the teacher is responsible for supplying all materials needed, as well as supervising:
· Here are some links for additional practice problems and worksheets:
· Because this material presents as very challenging to students, any consolidation activity should consist of more practice problems. Any of the previously suggested links can be used for consolidation.
References
- Image on top retrieved from: http://academic.pgcc.edu/~kroberts/Lecture/Chapter%205/05-02_RedoxReactions_L.jpg
- Di Guiseppe, M., Haberer, S., Salciccioli, K., Sanader, M, Vavitsas, A. (2012) Chemistry 12. Toronto: Nelson Education Ltd.
- van Kessel, H., Jenkins, F., Davies, L., Plumb, D., Di Guiseppe, M., Lantz, O., Tompkins, D. (2003). Chemistry 12. Toronto: Thomson Canada Limited