PREDICTING REDOX REACTIONS
Throughout the students’ academic life, the assumption has been made, explicitly or implicitly, that a reaction will occur if the reactants are mixed together. To the student, the balanced chemical equation for the combination of copper metal and silver nitrate solution looks as legitimate as the combination of copper metal and zinc nitrate solution. Yet one occurs spontaneously and the other does not.
This lesson provides an explanation for such discrepancy, and provides students with basic rules that can be used to predict the reactivity of redox reactions.
This lesson provides an explanation for such discrepancy, and provides students with basic rules that can be used to predict the reactivity of redox reactions.
Learning Objective
By the end of this lesson, I will:
· Be able to predict spontaneity of redox reactions using tables of oxidizing and reducing agents.
· Be able to predict spontaneity of redox reactions using tables of oxidizing and reducing agents.
Lesson Approaches and Overview (Specific Expectations: F2.1, F2.3, F3.1)
· As an introductory or hook activity, perform the “Spontaneity of Redox Reactions Activity” investigation presented in the following link:
The evidence collected in this mini-investigation can be used to synthesize some important generalizations on the reactivity of redox systems and to expand the theoretical understanding of these systems.
· It would be a good strategy to emphasize that the table of relative strengths of oxidizing and reducing agents created from the evidence obtained from investigations such as the mini-investigation performed is empirically developed, as supposed to being developed from redox theory.
· Using the CRC Handbook, use one of the half-reaction tables to estimate the number of half-reactions in it, and pass the book around to see the extensive investigations of reactions to build such a table.
· Any of the following videos can be watched, as they deal with predicting redox reactions:
· Using the CRC Handbook, use one of the half-reaction tables to estimate the number of half-reactions in it, and pass the book around to see the extensive investigations of reactions to build such a table.
· Any of the following videos can be watched, as they deal with predicting redox reactions:
· Watch the following tutorial about constructing a redox table:
Here is a link for an activity that would allow students to build a Redox table. The activity here suggested contains the answers, but the teacher can create a new document using the questions.
· Explain to students, that by convention, half-reaction equations are written as reductions. The strongest oxidizing agent (SOA) is at the top left in a table that of relative strengths of oxidizing and reducing agents and the strongest reducing agent (SRA) is at the bottom right of the table. The double arrows may indicate an equilibrium in some situations, but here they simply indicate that the half-reactions may be read from left to right (top arrow) or from right to left (bottom arrow).
· Have students work, in pairs or individually, on the following worksheet about predicting Redox reactions:
· Have students work, in pairs or individually, on the following worksheet about predicting Redox reactions:
· As part of the lecture, any of the following presentations can be used:
The following contains a few hints that are important for the prediction of redox reaction, which will help the students grasp the concepts in this lesson:
· Explain to students that elemental metal always lose electrons in redox reactions to form cations. As a result, metals are reducing agents. Conversely, metal cations usually gain electrons to form metal atoms. Hence, metal ions are usually oxidizing agents. Also explain that there are some exceptions. For example, the iron(II) ion can be both oxidized and reduced:
Oxidation: Fe2+(aq) ---> Fe3+(aq) + e-
Reduction: Fe2+(aq) + 2 e ---> Fe(s)
· Suggested lab: The reaction of sodium with water.
Oxidation: Fe2+(aq) ---> Fe3+(aq) + e-
Reduction: Fe2+(aq) + 2 e ---> Fe(s)
· Suggested lab: The reaction of sodium with water.
Once the experiment has been performed, have the students read and discuss the following described experiment called “The Great Sodium Disaster of 1998”:
· The following worksheet is a very good summary of the oxidation-reduction reactions, and it serves as a good consolidation activity:
· Students can research about bleach. Chlorine bleach has many applications as an oxidizing agent. It is used, among many other things, to remove hydrogen sulfide from groundwater. It also removes stains by oxidizing coloured substances to substances without colour. However, because of its reactivity with acids and bases, many workplaces have banned the use of chlorine bleach. Have students research the hazards of accidentally mixing chlorine bleach with acids and bases, uses of chlorine bleach, the use of hydrogen peroxide as a replacement for chlorine bleach, the contrast between the two, and any other safety precautions to be exercised when using chlorine bleach.
References
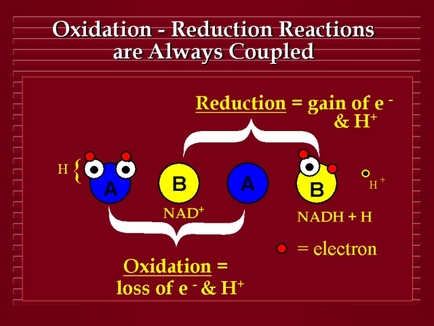
- Image on top retrieved from: http://quinton.pbworks.com/f/o.r.jpg
- Di Guiseppe, M., Haberer, S., Salciccioli, K., Sanader, M, Vavitsas, A. (2012) Chemistry 12. Toronto: Nelson Education Ltd.
- van Kessel, H., Jenkins, F., Davies, L., Plumb, D., Di Guiseppe, M., Lantz, O., Tompkins, D. (2003). Chemistry 12. Toronto: Thomson Canada Limited