STANDARD REDUCTION POTENTIALS
Learning Objectives
By the end of this lesson, students will:
· Be able to predict the cell potential for a galvanic cell using a reference table.
· Understand the concept of the standard cell potential.
· Relate cell potential to the activity series.
· Interpret the activity series in terms of elements that are more or less easily oxidized.
· Be able to predict the cell potential for a galvanic cell using a reference table.
· Understand the concept of the standard cell potential.
· Relate cell potential to the activity series.
· Interpret the activity series in terms of elements that are more or less easily oxidized.
Lesson Approaches and Overview (Specific Expectations: F1.1, F2.1, F2.3, F2.5, F2.6, F3.1, F3.2, F3.3, F3.4)
· Start by referring any of the galvanic cells created by students for this lesson.
· The following video is an excellent hook, and an excellent introduction to standard cell or reduction potentials:
· The following video is an excellent hook, and an excellent introduction to standard cell or reduction potentials:
· NOTE: Practicing cell potential problems is the most efficiently way to learn the content of this lesson. It is with that in mind that I provide a greater number of worksheet as teaching strategies.
· This lesson will deal with a bit amount of mathematics, so it is important that teachers take that into account when teaching cell potential, as referred to the students who find mathematics challenging.
· Remind the students that cell potential is a measure of the electric potential difference or voltage between two half- cells. A useful analogy is explaining t the students that cell potential is alike the difference in gravitational potential energy between the two apples hanging at different heights on the tree.
· Introduce the concept of volt, the SI unit for cell potential. Define volt in terms of joules and coulombs.
· A misconception that often arises can be debunked with the following explanation: cell potential is unaffected by the number of electrons transferred as a galvanic cell operates because cell potential is a ratio of energy to charge.
· Explain to the students that factors that influence cell potential include the design of the cell, the redox involved, and the concentration of the reactants in the cell.
· Define standard cell, standard cell potential and standard reduction potential. It is important that the formula:
ΔE0r(cell) = Eor(cathode) – Eor(anode)
is dissected and explained in details.
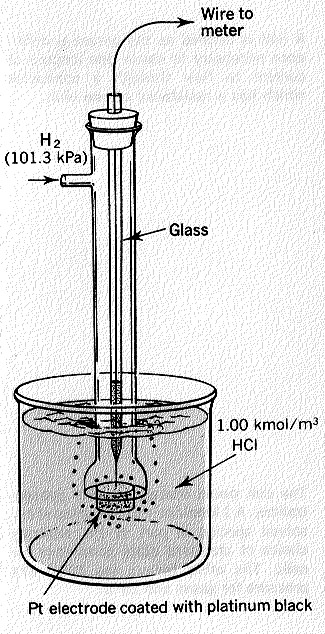
· Explain to students that in order to develop a reduction potentials table, a standard hydrogen half-cell is assigned an electric potential of exactly 0 volts. Define how hydrogen half-cells are constructed. Emphasize that although this reference half-cell potential is exactly zero volts, the value is often stated as 0.00 V to correspond to the measured potentials of the other half-cell potentials.
· This lesson will deal with a bit amount of mathematics, so it is important that teachers take that into account when teaching cell potential, as referred to the students who find mathematics challenging.
· Remind the students that cell potential is a measure of the electric potential difference or voltage between two half- cells. A useful analogy is explaining t the students that cell potential is alike the difference in gravitational potential energy between the two apples hanging at different heights on the tree.
· Introduce the concept of volt, the SI unit for cell potential. Define volt in terms of joules and coulombs.
· A misconception that often arises can be debunked with the following explanation: cell potential is unaffected by the number of electrons transferred as a galvanic cell operates because cell potential is a ratio of energy to charge.
· Explain to the students that factors that influence cell potential include the design of the cell, the redox involved, and the concentration of the reactants in the cell.
· Define standard cell, standard cell potential and standard reduction potential. It is important that the formula:
ΔE0r(cell) = Eor(cathode) – Eor(anode)
is dissected and explained in details.
· Explain to students that in order to develop a reduction potentials table, a standard hydrogen half-cell is assigned an electric potential of exactly 0 volts. Define how hydrogen half-cells are constructed. Emphasize that although this reference half-cell potential is exactly zero volts, the value is often stated as 0.00 V to correspond to the measured potentials of the other half-cell potentials.
- The following video is a good resource of an explanation of a hydrogen half-cell:
· The following video can be used during the lecture to either explain, or to reinforce all concepts in this lesson:
· These four videos are a series that presents a complete coverage of the lesson:
|
|
|
|
|
|
· The following websites provide very valuable information, and can be used during lectures:
· Emphasize to students that a reduction potential is like an elevation stated on a road map. Both represent a difference from a defined zero value-hydrogen reference half-cell for reduction potentials and sea level for elevations.
· The following mnemonics may help students remember the process that occurs at each electrode:
RED CAT – REDuction always occurs at the CAThode.
AN OX – OXidation always occurs at the ANode.
· The following is a good tip to help students determine the cathode and anode of a cell:
A voltmeter has two terminals, positive (red) and negative (black). Connect these to the electrodes of any cell so that the voltmeter gives a positive reading. Whatever electrode is connected to the positive terminal will be the cathode, and the other electrode will be the anode.
· After explaining through some examples, establish the relationship between the predicted cell potential and the spontaneity of the reaction:
1. Spontaneous reaction, ΔE0r(cell) = 0
A positive value for the cell potential means that the cell reaction occurs spontaneously.
2. Reaction at equilibrium , ΔE0r(cell) = 0
Eventually, the cell potential for an operating cell decreases to zero.
3. Non-spontaneous reaction, ΔE0r(cell) < 0
A negative value for the cell potential means that the cell reaction does not occur spontaneously.
· Use the Standard Reduction Potentials table to predict standard cell potentials. This is better achieved by having students spend a significant amount of time practicing.
· The following mnemonics may help students remember the process that occurs at each electrode:
RED CAT – REDuction always occurs at the CAThode.
AN OX – OXidation always occurs at the ANode.
· The following is a good tip to help students determine the cathode and anode of a cell:
A voltmeter has two terminals, positive (red) and negative (black). Connect these to the electrodes of any cell so that the voltmeter gives a positive reading. Whatever electrode is connected to the positive terminal will be the cathode, and the other electrode will be the anode.
· After explaining through some examples, establish the relationship between the predicted cell potential and the spontaneity of the reaction:
1. Spontaneous reaction, ΔE0r(cell) = 0
A positive value for the cell potential means that the cell reaction occurs spontaneously.
2. Reaction at equilibrium , ΔE0r(cell) = 0
Eventually, the cell potential for an operating cell decreases to zero.
3. Non-spontaneous reaction, ΔE0r(cell) < 0
A negative value for the cell potential means that the cell reaction does not occur spontaneously.
· Use the Standard Reduction Potentials table to predict standard cell potentials. This is better achieved by having students spend a significant amount of time practicing.
· It is a good strategy to summarize, and have the students write down, the rules for analyzing standard cells. These rules can be broken down as follows:
1. The cathode is the electrode where the strongest oxidizing agent present in the cell reacts, i.e., the oxidizing agent in the cell that is closest to the top on the left side of the redox table. If required, copy the reduction half-reaction for the strongest oxidizing agent and its reduction potential.
2. The node is the electrode where the strongest reducing agent present in the cell reacts, i.e., the reducing agent in the cell that is closest to the bottom on the right side of the redox table. If required, copy the oxidation half-reaction (reverse the half-reaction by reading from right to left) for the strongest reducing agent and the reduction potential listed on the table.
3. Balance the electrons for the two half-reactions (but do not change the Ers) and add the half-reaction equations to obtain the overall or net cell reaction.
4. Calculate the standard cell potential ΔE0.
· The following are some suggested worksheets:
1. The cathode is the electrode where the strongest oxidizing agent present in the cell reacts, i.e., the oxidizing agent in the cell that is closest to the top on the left side of the redox table. If required, copy the reduction half-reaction for the strongest oxidizing agent and its reduction potential.
2. The node is the electrode where the strongest reducing agent present in the cell reacts, i.e., the reducing agent in the cell that is closest to the bottom on the right side of the redox table. If required, copy the oxidation half-reaction (reverse the half-reaction by reading from right to left) for the strongest reducing agent and the reduction potential listed on the table.
3. Balance the electrons for the two half-reactions (but do not change the Ers) and add the half-reaction equations to obtain the overall or net cell reaction.
4. Calculate the standard cell potential ΔE0.
· The following are some suggested worksheets:
|
|
|
| ||||||||||||||||||||||||
· As consolidation ideas, any of the worksheets presented above would be efficient.
· This lesson introduced hydrogen cells. Students, therefore, can be asked to investigate the technology behind cars that use hydrogen fuel cells. Ask students to describe the practical problems of hydrogen fuel cell as a source of energy in the transportation section. This research can be done as homework, or during class periods. Students should be encouraged to present their results to the class as a whole.
· This lesson introduced hydrogen cells. Students, therefore, can be asked to investigate the technology behind cars that use hydrogen fuel cells. Ask students to describe the practical problems of hydrogen fuel cell as a source of energy in the transportation section. This research can be done as homework, or during class periods. Students should be encouraged to present their results to the class as a whole.
References
- Image on top of retrieved from: http://resources.educ.queensu.ca/science/main/concept/chem/c12/Eric/Grade%2011%20Electrochemistry/images/galvanic_cell_b.jpg
- Di Guiseppe, M., Haberer, S., Salciccioli, K., Sanader, M, Vavitsas, A. (2012) Chemistry 12. Toronto: Nelson Education Ltd.
- van Kessel, H., Jenkins, F., Davies, L., Plumb, D., Di Guiseppe, M., Lantz, O., Tompkins, D. (2003). Chemistry 12. Toronto: Thomson Canada Limited