ELECTROLYSIS AND ELECTROLYTIC CELLS
By this point in the Electrochemistry unit, students are familiar with all the concepts needed to explain electrolysis. The concepts that are common to all electrochemical cells, galvanic and electrolytic, are:
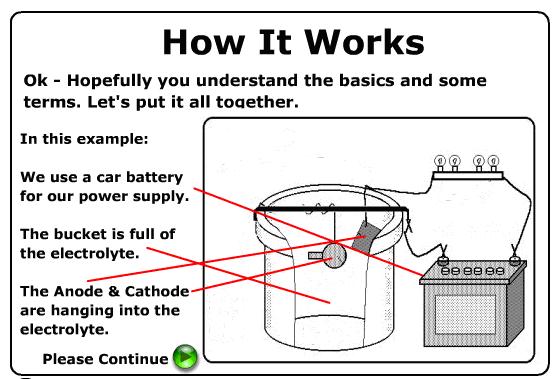
· An electrochemical cell always contains two electrodes and at least one electrolyte.
· In an electrochemical cell, oxidation occurs at the anode and reduction occurs at the cathode.
· In an operating electrochemical cell, anions migrate towards the anode, and cations migrate toward the cathode.
· In an operating electrochemical cell, electrons always move from the anode (where electrons are lost through oxidation) to the cathode (where electrons are gained through reduction).
· Standard cell potential difference can be predicted from a table of reduction potentials.
The differences between the two types of cells are:
· Electrochemical cell reactions can be spontaneous or non spontaneous.
· Electrochemical cell potential differences can be positive or negative.
· The cathode can be labeled positive or negative.
· An electrochemical cell always contains two electrodes and at least one electrolyte.
· In an electrochemical cell, oxidation occurs at the anode and reduction occurs at the cathode.
· In an operating electrochemical cell, anions migrate towards the anode, and cations migrate toward the cathode.
· In an operating electrochemical cell, electrons always move from the anode (where electrons are lost through oxidation) to the cathode (where electrons are gained through reduction).
· Standard cell potential difference can be predicted from a table of reduction potentials.
The differences between the two types of cells are:
· Electrochemical cell reactions can be spontaneous or non spontaneous.
· Electrochemical cell potential differences can be positive or negative.
· The cathode can be labeled positive or negative.
Learning Objectives
By the end of this lesson, the students will:
· Be able to identify environmental implications of industrial electrochemical processes.
· Understand, and be able to explain, the concept of electrolysis as it relates to redox reactions.
· Identify the similarities and differences between electrolytic and galvanic cells.
· Be able to describe some of the industrial applications of electrolysis.
· Be able to identify environmental implications of industrial electrochemical processes.
· Understand, and be able to explain, the concept of electrolysis as it relates to redox reactions.
· Identify the similarities and differences between electrolytic and galvanic cells.
· Be able to describe some of the industrial applications of electrolysis.
Lesson Approaches and Overview (Specific Expectations: F1.2, F2.1, F2.3, F3.1, F3.5)
· The following link is an appropriate video to introduce the concept of electrolysis. It is a catchy, fun song about electrolysis:
Here are some other videos that can be used as introduction to the lesson, or hooks:
|
|
|
|
|
|
· Students can be asked to come up with the timeline, or chronology of electrolysis. This would provide a good illustration of how a new technology can facilitate scientific research
· It is important to emphasize that certain electrochemical reactions are non-spontaneous.
· Some students have difficulty distinguishing between galvanic and electrolytic cells. Emphasize that in a galvanic cell a spontaneous redox reaction is used to produce electricity, while in an electrolytic cell an external source of electricity is used to force a non-spontaneous reaction to occur.
· Emphasize that in galvanic cells the anode is labeled negative and the cathode is positive, while it is the other way around an electrolytic cell.
· Remind students that all electrochemical reactions can be described theoretically in the same way: list entities present, write the half-reactions for the strongest oxidizing agent and the strongest reducing agent, and writing the net reaction equation.
· A useful learning tip is using the mnemonic:
SOAC GERC (“soak a jerk”): the Strongest Oxidizing Agent at the Cathode Gains Electrons and is Reduced at the Cathode.
· The following are resources that can be used during the lecturing part of the lesson:
· It is important to emphasize that certain electrochemical reactions are non-spontaneous.
· Some students have difficulty distinguishing between galvanic and electrolytic cells. Emphasize that in a galvanic cell a spontaneous redox reaction is used to produce electricity, while in an electrolytic cell an external source of electricity is used to force a non-spontaneous reaction to occur.
· Emphasize that in galvanic cells the anode is labeled negative and the cathode is positive, while it is the other way around an electrolytic cell.
· Remind students that all electrochemical reactions can be described theoretically in the same way: list entities present, write the half-reactions for the strongest oxidizing agent and the strongest reducing agent, and writing the net reaction equation.
· A useful learning tip is using the mnemonic:
SOAC GERC (“soak a jerk”): the Strongest Oxidizing Agent at the Cathode Gains Electrons and is Reduced at the Cathode.
· The following are resources that can be used during the lecturing part of the lesson:
|
|
|
|
|
|
· Some PowerPoint presentation that can be used as a reference guide are included here:
· Here are some suggested worksheets that can be used as formative assessment, or simply to reinforce the taught concepts:
· For the part of the lecture that highlights the differences and similarities between galvanic and electrolytic cells, here are some suggestions:
The following have a table that compares galvanic versus electrolytic cells. The teacher is encouraged to ask students to create their own tables as an in-class activity:
Here are one suggested lesson plan and one suggested PowerPoint presentation about the differences between the two electrochemical cells:
|
| ||||||||||||
· This lesson benefits greatly from performing hands-on experiments. Here are my suggestions for good, effective experiments:
· For the part of the lesson that deals with the electrolysis of water, these videos could be useful:
|
|
|
|
|
|
INDUSTRIAL USES OF ELECTROLYSIS:
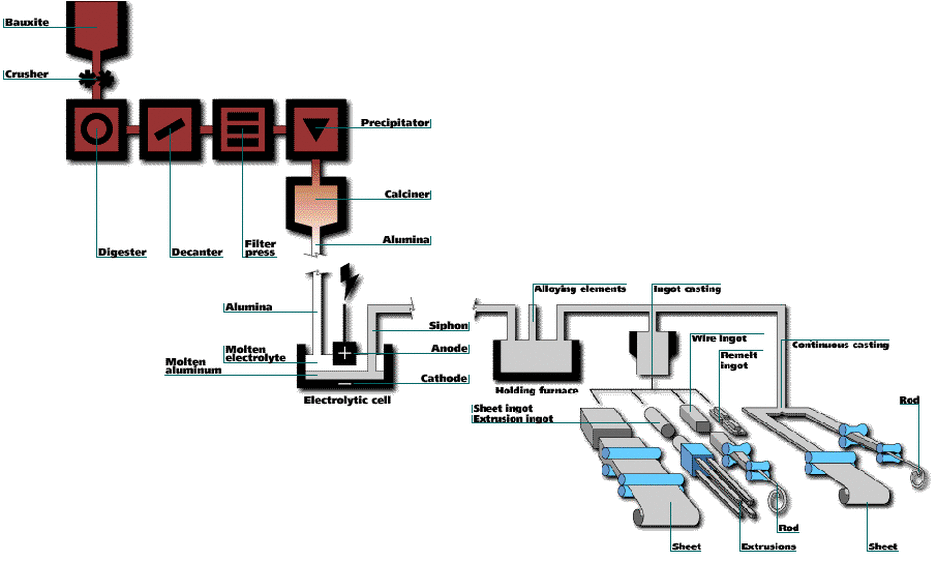
· Introduce this part of the lesson by explaining to the students that the production of aluminum is important to Canada’s economy, although Canada does not have large deposits of aluminum ore. Explain that hydroelectric power is used to produce aluminum metal from concentrated bauxite in an electrolytic cell. Emphasize that the recycling of aluminum from soft drinks and beer cans requires only 5% of the energy required to produce aluminum by electrolysis.
· Introduce this part of the lesson by explaining to the students that the production of aluminum is important to Canada’s economy, although Canada does not have large deposits of aluminum ore. Explain that hydroelectric power is used to produce aluminum metal from concentrated bauxite in an electrolytic cell. Emphasize that the recycling of aluminum from soft drinks and beer cans requires only 5% of the energy required to produce aluminum by electrolysis.
- Here is a general video about the applications of electrolysis:
· Here are some interesting videos about the production of aluminum:
|
|
|
An interesting fact about aluminum is that the five largest aluminum refineries in the Western world are located in Canada. Relate this to the fact that, per capita, Canadians use more energy per capita than citizen of other countries.
Students can be assigned the task to find out all the information about the refineries (where they are located, how many people they employ, etc.). To tie in this theme with the curriculum expectations, students should research the environmental issues associated with aluminum refineries.
Here are 4 websites with very good general information about the production of aluminum:
Students can be assigned the task to find out all the information about the refineries (where they are located, how many people they employ, etc.). To tie in this theme with the curriculum expectations, students should research the environmental issues associated with aluminum refineries.
Here are 4 websites with very good general information about the production of aluminum:
· Another industrial application of electrolysis is the electrolysis of sodium chloride solution. Explain to the students that one of the uses of sodium metal is the production of sodium vapour lamps used for street lighting. Sodium lamps produce a yellow light that penetrates farther than white light and allows better vision in fog. Sodium lamps are also fifteen time more efficient that regular incandescent lamps.
Here are a video about the electrolysis of salt, and the chloralkali process:
Here are a video about the electrolysis of salt, and the chloralkali process:
|
|
|
The following are reference websites about the chloralkali process:
The following website is an interactive website about the electrolysis of sodium chloride:
· Here is a link to another application of electrolysis, the removal of rust:
· A very important use of electrolysis is the electrorefining of metals. Here are some valuable links for this topic:
|
|
|
· Another use of electrolysis is the process of electroplating. Explain to the students that the Electroplating the Canadian dollar coin is a very typical example of how a form of technology came to pass-the optimum conditions for electroplating the dollar coin were determined by trial and error. Emphasize that to achieve the best compromise among the expensive prices of gold, zinc and chromium, their mechanical properties, appearance, and corrosion resistance, utensils or jewelry made by made of a relatively inexpensive, yet string, allot, and then coated with another metal or alloy to improve appearance or corrosion resistance. This process is known as electroplating.
Here are links to videos and websites that do a great job at explaining electroplating. Note that the last YouTube is an animation.
Here are links to videos and websites that do a great job at explaining electroplating. Note that the last YouTube is an animation.
|
|
|
- Any of the activities, worksheets, videos can be used as debrief/consolidation.
References
- Image on top retrieved from: http://images.flatworldknowledge.com/averill/averill-fig19_020.jpg
- Di Guiseppe, M., Haberer, S., Salciccioli, K., Sanader, M, Vavitsas, A. (2012) Chemistry 12. Toronto: Nelson Education Ltd.
- van Kessel, H., Jenkins, F., Davies, L., Plumb, D., Di Guiseppe, M., Lantz, O., Tompkins, D. (2003). Chemistry 12. Toronto: Thomson Canada Limited