QUANTITATIVE CHANGES IN EQUILIBRIUM-REACTION QUOTIENTS
What is a Reaction Quotient (Q)?
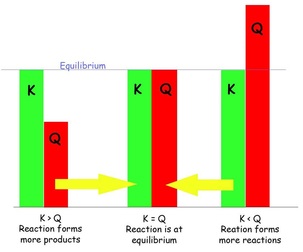
Simply put reaction quotients are a mathematical tool used to determine whether or not a system is at equilibrium and if not, which way the system must react or "shift" to achieve equilibrium. The calculation for the reaction quotient is the same one that is used to determine the equilibrium constant, K, the only difference being that the concentrations inputted into the mathematical expression for K are equilibrium concentrations whereas the concentrations used for the reaction quotient, Q, are which ever concentrations are present or given.
The introduction of reaction quotients is an extension of the both the knowledge of equilibrium constants and of Le Chatelier's Principle. It is essentially introducing the mathematical consequences of Le Chatelier's Principle.
Simply put reaction quotients are a mathematical tool used to determine whether or not a system is at equilibrium and if not, which way the system must react or "shift" to achieve equilibrium. The calculation for the reaction quotient is the same one that is used to determine the equilibrium constant, K, the only difference being that the concentrations inputted into the mathematical expression for K are equilibrium concentrations whereas the concentrations used for the reaction quotient, Q, are which ever concentrations are present or given.
The introduction of reaction quotients is an extension of the both the knowledge of equilibrium constants and of Le Chatelier's Principle. It is essentially introducing the mathematical consequences of Le Chatelier's Principle.
LESSON OVERVIEW (SPECIFIC EXPECTATIONS E2.1, E2.2, E2.4)
Lesson Hook:
The calculation of reaction quotients and the resulting relation to the equilibrium constant requires a firm grasp of mathematical concepts such as ratios, the quadratic equation, isolation of variables and "completing the square"; as well as a strong understanding of the qualitative principles behind Le Chatelier's Principle. As such, it may be a good idea to review both Le Chatelier's Principle and these mathematical concepts before introducing reaction quotients. This can be used as a mini-diagnostic to determine the level of mathematical comprehension of the class before going forward. It may be necessary to allow students to practice these skills such that when the concept of reaction quotients is introduced, the mathematical portion does not distract them from the new concept being introduced. Click below for practice worksheets on completing the square and the quadratic formula courtesy of www.MathWorksheetsGo.com
The calculation of reaction quotients and the resulting relation to the equilibrium constant requires a firm grasp of mathematical concepts such as ratios, the quadratic equation, isolation of variables and "completing the square"; as well as a strong understanding of the qualitative principles behind Le Chatelier's Principle. As such, it may be a good idea to review both Le Chatelier's Principle and these mathematical concepts before introducing reaction quotients. This can be used as a mini-diagnostic to determine the level of mathematical comprehension of the class before going forward. It may be necessary to allow students to practice these skills such that when the concept of reaction quotients is introduced, the mathematical portion does not distract them from the new concept being introduced. Click below for practice worksheets on completing the square and the quadratic formula courtesy of www.MathWorksheetsGo.com
|
| ||||||||||||
Action:
This topic within the systems in equilibrium concept is very heavy in calculations, making it difficult to maintain a level of interest in the classroom. A good idea to try is to get a little game of "determine that reaction quotient" going after introducing the concept and its relation to Le Chatelier's Principle. For this game, students are grouped (no more than 4 ideally) into teams. Each team is given a mini whiteboard or some other means of displaying the answer. The teacher writes a question on the board and each member of each team attempts to find the correct answer. Each team then agrees on one answer and displays it in the air. The first team with the correct answer gets one point-with one bonus point going to the team that can correctly identify which way the system must shift to reach equilibrium. The team with the most points at the end of the game wins the opportunity to create the homework question for the other teams (note: they must also do the question to determine the correct answer)-and a lollipop if so desired by the teacher. This game is a great tool for a formative assessment as well, as the teacher can gauge student understanding by both the amount of answers correct as well as the way in which students gave incorrect answers.
This topic within the systems in equilibrium concept is very heavy in calculations, making it difficult to maintain a level of interest in the classroom. A good idea to try is to get a little game of "determine that reaction quotient" going after introducing the concept and its relation to Le Chatelier's Principle. For this game, students are grouped (no more than 4 ideally) into teams. Each team is given a mini whiteboard or some other means of displaying the answer. The teacher writes a question on the board and each member of each team attempts to find the correct answer. Each team then agrees on one answer and displays it in the air. The first team with the correct answer gets one point-with one bonus point going to the team that can correctly identify which way the system must shift to reach equilibrium. The team with the most points at the end of the game wins the opportunity to create the homework question for the other teams (note: they must also do the question to determine the correct answer)-and a lollipop if so desired by the teacher. This game is a great tool for a formative assessment as well, as the teacher can gauge student understanding by both the amount of answers correct as well as the way in which students gave incorrect answers.
Debrief:
To debrief after the game, a good idea would be to go through a question or two that the majority of the teams had difficulty with-reinforcing the concept with a worked example and stressing the concept that the equilibrium constant is a special circumstance of the more general reaction quotient.
FOPP: Commitment to students and student learning and professional practice.
To debrief after the game, a good idea would be to go through a question or two that the majority of the teams had difficulty with-reinforcing the concept with a worked example and stressing the concept that the equilibrium constant is a special circumstance of the more general reaction quotient.
FOPP: Commitment to students and student learning and professional practice.
The concept of reaction quotients can be reinforced in a variety of ways to appeal to all forms of multiple intellegences. Click below for a few resources that can be used in conjunction with a lesson to further demonstrate the concept of reaction quotients