SOCIETAL IMPLICATIONS AND APPLICATIONS OF ELECTROCHEMISTRY
The uses of electrochemistry are wide and varied. Electrochemistry plays a very significant role in our daily lives, and thus, its societal implications and applications are an intrinsic part of our society's fabric. Here is a list of some of the its most common uses and implications:
BIOMEDICAL APPLICATIONS
Various metallic or non-metallic surfaces are frequently treated by electrochemical methods in order to achieve desirable properties important to biomedical applications. Applications include:
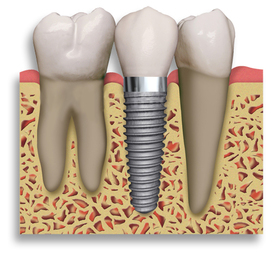
· Orthopedic and dental implants
· Dressings for wound healing and different skin dressings
· Surfaces for the prevention of bio-film formation or corrosion inhibition in biological media (implants, etc.)
· Certain diabetes blood sugar meters the amount of glucose in the blood through its redox potential.
· Orthopedic and dental implants
· Dressings for wound healing and different skin dressings
· Surfaces for the prevention of bio-film formation or corrosion inhibition in biological media (implants, etc.)
· Certain diabetes blood sugar meters the amount of glucose in the blood through its redox potential.
|
|
The following video explains the fabrication of an orthopedic implant:
METALLURGIC PROCESSES
· Electrochemistry is used in the production and extraction of metals like aluminum and titanium. It represents the most economically viable option of aluminum production in Canada.
· Metals like copper, zinc, silver and lead are refined by electrochemistry.
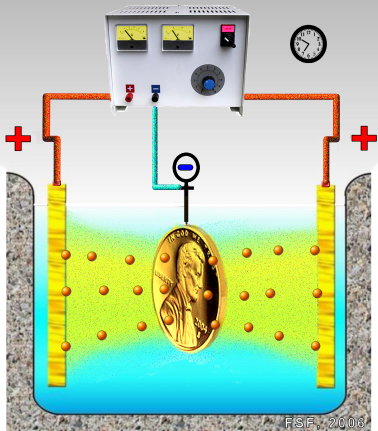
· The process of electroplating is used to protect metal articles from corrosion by coating them with a more corrosion-resistant metal. As expected, the prevention of corrosion has many important implications for our society.
· A special application of electroplating is electroforming. In electroforming, a prefabricated form is completely filled with a thick metal deposit (a reduction process) to produce a solid metal object; no further machining is needed to obtain the final object. This is the process used to form precious metal jewelry.
· Decorative coatings are also applied electrochemically. Utensils and jewelry are just two of the many examples of this application.
· Metals like copper, zinc, silver and lead are refined by electrochemistry.
· The process of electroplating is used to protect metal articles from corrosion by coating them with a more corrosion-resistant metal. As expected, the prevention of corrosion has many important implications for our society.
· A special application of electroplating is electroforming. In electroforming, a prefabricated form is completely filled with a thick metal deposit (a reduction process) to produce a solid metal object; no further machining is needed to obtain the final object. This is the process used to form precious metal jewelry.
· Decorative coatings are also applied electrochemically. Utensils and jewelry are just two of the many examples of this application.
|
|
The following videos are about the process by which aluminum is made. The second video includes the fabrication of snow boards, something some students may find interesting:
|
|
|
The following videos are about the process of making gold jewelry, and gold electroplating respectively:
|
|
|
The following videos are about the process of electroforming for copper and jewelry, respectively:
|
|
|
THE PRODUCTION OF CHLORINE
· Electrochemistry is used to produce chlorine through the electrolysis of brine (the bases of the chlor-alkali process-See Glossary).
· Chlorine plays an important part in our daily lives. In many parts of the world, drinking water is treated with chlorine.
· Chlorine is a basic ingredient in many plastics like PVC, which is used for piping.
· Many cleaning agents, detergents and soaps are made with the caustic-soda that is generated in the brine-electrolysis cells.
· Chlorine plays an important part in our daily lives. In many parts of the world, drinking water is treated with chlorine.
· Chlorine is a basic ingredient in many plastics like PVC, which is used for piping.
· Many cleaning agents, detergents and soaps are made with the caustic-soda that is generated in the brine-electrolysis cells.
The following videos are about the chlor-alkali process (that makes chlorine), and about how PVC gloves are made
|
|
|
ENVIRONMENTAL IMPLICATIONS
· Electrochemical techniques have proven to be a clean, flexible and powerful tool for the development of new methods for waste and water treatment.
· Electrochemistry is the process behind the production of batteries currently used in “hybrid cars.” These batteries are viable candidates for power source in fully electric cars.
· In another version of the electric car, the power could be provided by “fuel cells”, which are batteries that can be continuously fed by a fuel. However, the cost of these cells has rendered this technological use prohibitive thus far.
· Electrochemical processes are often used in environment protection methods.
· Electrochemistry is the process behind the production of batteries currently used in “hybrid cars.” These batteries are viable candidates for power source in fully electric cars.
· In another version of the electric car, the power could be provided by “fuel cells”, which are batteries that can be continuously fed by a fuel. However, the cost of these cells has rendered this technological use prohibitive thus far.
· Electrochemical processes are often used in environment protection methods.
The following videos are excellent resources about hydrogen fuel cells:
|
|
|
BATTERIES
· Perhaps one of the most important uses of electrochemistry, and by far the most widely appreciated, is the production of batteries. To appreciate how important the existence, development and maintenance of batteries, one has to look no further than our own bags, wrists, households, cars, etc. The simple “throw away batteries’, those used in toy cars, wireless keyboards, etc., are electrochemical cells.
· The more sophisticated version of batteries permits for the batteries be re-used by recharging with electricity, a money-saving advantage.
· The more sophisticated version of batteries permits for the batteries be re-used by recharging with electricity, a money-saving advantage.
The following videos are about how batteries are made. These videos are very interesting and provide a great resources to "hook" students to electrochemistry:
|
|
|
OTHER "LESS-THOUGHT-OF" APPLICATIONS OF ELECTROCHEMISTRY
· In animal and plant life: photosynthesis and respiration are inherently electrochemical processes.
· Electrochemical processes are often used to clean and restore archeological artifacts.
· The production if some electrical circuit components often involves electrochemical processes.
· In addition to fuel cells and batteries already in use in space programs, electrochemical processes have been also proposed for outer-space applications.
· Electrochemical processes are often used to clean and restore archeological artifacts.
· The production if some electrical circuit components often involves electrochemical processes.
· In addition to fuel cells and batteries already in use in space programs, electrochemical processes have been also proposed for outer-space applications.
|
|
The following websites are valuable resources to use when teaching about the applications of electrochemistry:
References
- http://www.wikipedia.org/
- http://www.ingentaconnect.com/content/ben/eng/2007/00000001/00000003/art00007
- http://electrochem.cwru.edu/encycl/art-i02-introduction.htm
- http://m.everythingscience.co.za/grade-12/04-electrochemical-reactions/04-electrochemical-reactions-06.cnxmlplus
- DiGiuseppe, M., Haberer, S., Salciccioli, K., Sanader, M., & Vavitsas, A. (2012). Nelson Chemistry 12 University preparation. Toronto, ON: Nelson Education Ltd.
- van Kessel, H., Jenkins, F., Davies, L., Plumb, D., Di Guiseppe, M., Lantz, O., Tompkins, D. (2003). Chemistry 12. Toronto: Thomson Canada Limited