Three-Dimensional Structure
8C
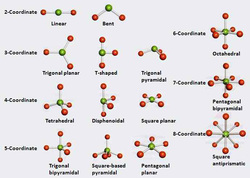
This refers to the three-dimensions of the molecule that is formed by combining atoms. The Valence Shell Electron-Pair Repulsion (VSEPR) theory predicts the distribution of atoms bonded to a central atom. VSEPR can predict the shapes and angles that molecules will take based on bonding and lone electrons.
Hook:
Enzymes are molecules that help speed up reactions without actually being used up in the reactions themselves. This fun video shows how the importance of molecular shape is essential for function.
Students will complete VESPR origami shapes that represent different configurations. See external resources for more information.
Consolidate:
Teacher will right a chemical formula on the board, and students must hold up the corresponding paper origami shape.
