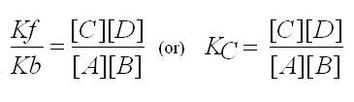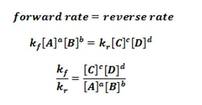EQUILIBRIUM LAW IN CHEMICAL SYSTEMS
What is Equilibrium Law in Chemical Systems?
Unlike previous reactions introduced, a system in equilibrium never proceeds to completion-there is always a mixture of reactants and products at equilibrium. In the mid-1800's two scientists, Guldberg and Waage, determined that if equilibrium concentrations were arranged in a specific mathematical ratio, the resulting value was constant, regardless of initial starting concentrations. They called this the "equilibrium law" and the resulting value the "equilibrium constant".
Unlike previous reactions introduced, a system in equilibrium never proceeds to completion-there is always a mixture of reactants and products at equilibrium. In the mid-1800's two scientists, Guldberg and Waage, determined that if equilibrium concentrations were arranged in a specific mathematical ratio, the resulting value was constant, regardless of initial starting concentrations. They called this the "equilibrium law" and the resulting value the "equilibrium constant".
LESSON OVERVIEW (SPECIFIC EXPECTATIONS: E2.1, E2.4, E3.2)
Lesson Hook:
At the beginning of this lesson, ask students to write down their definitions of the words "law" and "constant" and then share these definitions with the class. This gets them thinking of the meaning of those words as you ask them what they think as a class the terms "equilibrium law" and "equilibrium constant" means.
At the beginning of this lesson, ask students to write down their definitions of the words "law" and "constant" and then share these definitions with the class. This gets them thinking of the meaning of those words as you ask them what they think as a class the terms "equilibrium law" and "equilibrium constant" means.
Action:
Once this short thought activity is completed, the best way for students to see how the equilibrium constant works is to provide them with the mathematical proof. It reminds them of the definition of equilibrium and gives them a mathematical reason for the form the equilibrium constant takes and puts it into context. Deriving the constant also helps when Le Chatelier's principle is introduced and allows for those with weaker mathematical skills to have the opportunity for reinforcement.
Proof:
While going through the proof, remind students of the definition of equilibrium and highlight that "at equilibrium, the rate of the forward reaction equals the rate of the reverse reaction, then state the exact same thing while writing it mathematically as written below as many students find it difficult to translate words into mathematical expressions.
Once this short thought activity is completed, the best way for students to see how the equilibrium constant works is to provide them with the mathematical proof. It reminds them of the definition of equilibrium and gives them a mathematical reason for the form the equilibrium constant takes and puts it into context. Deriving the constant also helps when Le Chatelier's principle is introduced and allows for those with weaker mathematical skills to have the opportunity for reinforcement.
Proof:
While going through the proof, remind students of the definition of equilibrium and highlight that "at equilibrium, the rate of the forward reaction equals the rate of the reverse reaction, then state the exact same thing while writing it mathematically as written below as many students find it difficult to translate words into mathematical expressions.
To ensure that students understand the relationship between species concentration and the equilibrium constant, including how a change in one inherantly affects the other, have students investigate this relationship using the computer simulation suggested below curtesy of the Wolfram demonstrations project. Students can play with reactant and product concentration as well as alter the magnitude of the equilbrium constant and view the resulting changes in concentrations. Please click below to be redirected to the Wolfram demonstration-The Law of Mass Action.
Debrief:
Allow students to practice this new concept in class by incorporating equilibrium constant calculations into I.C.E. calculations done previously. This allows them to see the continuity of the lessons and concepts and reinforces previous work.
FOPP: Commitment to students and student learning, professional knowledge and ongoing professional learning
Allow students to practice this new concept in class by incorporating equilibrium constant calculations into I.C.E. calculations done previously. This allows them to see the continuity of the lessons and concepts and reinforces previous work.
FOPP: Commitment to students and student learning, professional knowledge and ongoing professional learning
The concept of the law of mass action can be reinforced in a variety of ways to appeal to all forms of multiple intellegences. Click below for a few resources that can be used in conjunction with a lesson to further demonstrate the concept of equilibrium law in chemical systems.